|
These electron-carrying molecules will shuttle the electrons to the ETC for oxidative phosphorylation.Īs the electrons are passed along the chain, every ETC member undergoes a redox reaction, accepting and donating electrons. Through redox reactions, most of the high-energy electrons are transferred to NAD + and FAD to create NADH (and H +) and FADH 2, respectively. Most of the ATP will come from oxidative phosphorylation, which will take place at the mitochondrial membrane where the electron transport chain (ETC) and the enzyme ATP synthase are embedded. The pyruvate from glycolysis is converted into acetyl CoA that will enter the mitochondrion for complete oxidation and degradation into carbon dioxide.įor every pyruvate molecule, the citric acid cycle will generate one ATP via substrate phosphorylation. The matrix is the location of the citric acid cycle, a cyclic metabolic reaction where food molecules are churned to generate energy-rich phosphate compounds. The space within the inner membrane is called the mitochondrial matrix. 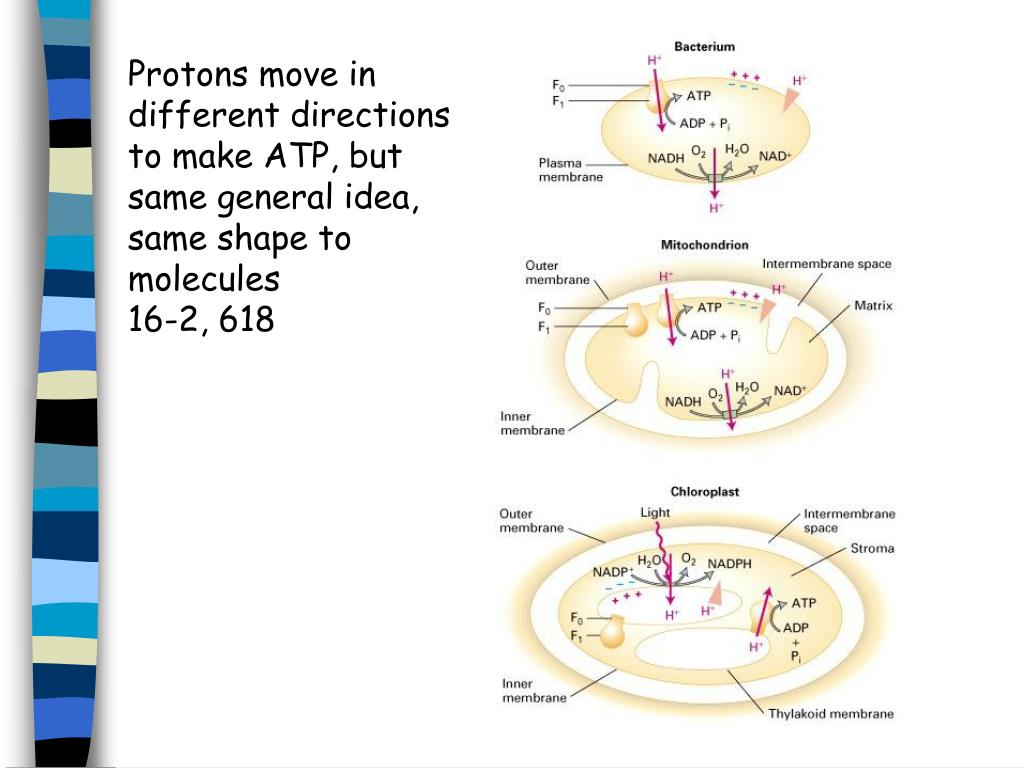
The inner membrane forms many infoldings called cristae. In between the two membranes is the intermembrane space. Both layers consist of lipid layers that prohibit the easy passage of ions. The mitochondrial membrane is made up of an outer membrane and an inner membrane. 
Take note that this organelle is a double-membraned structure. It is regarded as the powerhouse of the cell because most ATPs are produced here. The figure above is a schematic diagram of the mitochondrion. A chemiosmotic coupling model in the mitochondrion. To further explain the process of chemiosmosis and describe how it is a part of cellular respiration, see the diagram below. In respiring cells, it is one of the major steps of cellular respiration. Chemiosmosis ModelĬhemiosmosis is an energy-coupling mechanism employed by living organisms to produce ATP. This provided the basis as to how oxidative phosphorylation led to ATP synthesis. He showed that ATP synthesis was coupled to an electrochemical proton gradient. Mitchell proposed that ATP could also be produced by chemiosmosis. How these organelles generate ATP was initially not clear and was presumed to relate to substrate-level phosphorylation (as what happens in glycolysis). The mitochondria have long been known as the organelles responsible for ATP synthesis. However, how living organisms produce ATP biologically was not well established. 1) ATP was also already recognized at that time as the cell’s major energy currency. In the 1960s, he knew about the phenomenon of membrane potential in which the inner side of the membrane being negative relative to its environment. The discovery of ATP synthase and the pH difference across the thylakoid had the bioenergetics community consider the validity of his hypothesis. Mitchell’s hypothesis, however, was not accepted instantly until a substantive groundwork on proton pumping was laid. Mitchell (1920 – 1992), a British biochemist. Prokaryotes lack these organelles and therefore chemiosmosis will occur in their cell membrane.Īccording to the chemiosmotic theory, chemiosmosis is driven by an electrochemical proton gradient essential during the production of ATP. W here does chemiosmosis occur? In eukaryotes, it occurs in the mitochondria during cellular respiration and in the chloroplasts during photosynthesis. In chemiosmosis, the formation of an ion gradient leads to the generation of potential energy that is sufficient to drive the process. ATP) as opposed to an active transport system that does. Furthermore, it does not require chemical energy (e.g. Chemiosmosis uses membrane proteins to transport specific ions. These proteins in the membrane facilitate their movement by acting as a temporary shuttle or by serving as a channel or a passageway. Membrane proteins help the ions to move across since the membrane is not readily permeable to ions, basically because of its bilipid feature. Also, the molecules are transferred to the other side of the membrane with the help of membrane proteins. It is also similar to other forms of passive transport, such as facilitated diffusion. Not only is chemiosmosis similar to osmosis. As for chemiosmosis, the movement of ions is driven by an electrochemical gradient, such as a proton gradient. The differences in the pressures between the two sides of the membrane drive osmosis. In osmosis, this is referred to as an osmotic gradient. Nevertheless, both processes require a gradient. In the case of chemiosmosis, though, it involves the ions moving across the membrane in osmosis, it is the water molecules. 
The tendency for movement in this way is much like water accumulated on one side of a dam, moving through the dam when opened.This process is similar to osmosis where water molecules move passively.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
